To reduce thromboembolic risk, resume anticoagulant therapy as soon as medically appropriate following treatment with AndexXa ®.įDA gave this drug an accelerated approval and is the only approved product for reversal of apixaban and rivaroxaban. Also, monitor for symptoms and signs that precede cardiac arrest and provide treatment as needed. The package insert recommends to monitor for thromboembolic events and initiate anticoagulation when medically appropriate. 
Ischemic events, including myocardial infarction and ischemic stroke.Arterial and venous thromboembolic events.The side effect profile is low, however andexanet does carry the following warning that the drug is associated with serious and life-threatening adverse events, including: 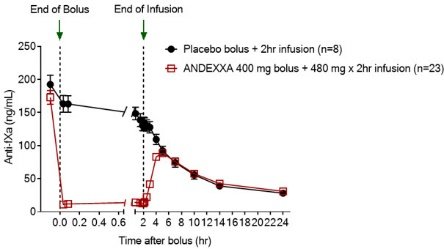
The package insert for AndexXa ® states an improvement in hemostasis has not been established. The completed trial in healthy volunteers and ongoing research trial with patients with acute major bleeds show overall efficacy in the agent’s ability to decrease anti-FXa activity. In addition, it inhibits the activity of Tissue Factor Pathway Inhibitor (TFPI), increasing tissue factor-initiated thrombin generation. It works as a reversal agent by binding and sequestering Factor Xa inhibitors. 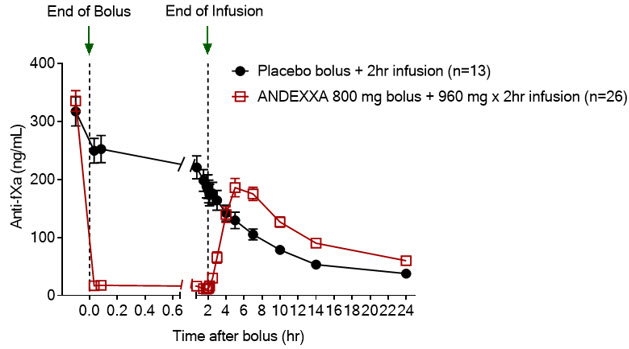
Andexanet alfa is a recombinant modified human Factor Xa protein that is catalytically inactive. Aandexanet alfa is indicated for patients who have been treated with rivaroxaban (Xarelto ®) or apixaban (Eliquis ®), and are in need of anticoagulation reversal due to life-threatening or uncontrolled bleeding. The first Factor Xa inhibitor reversal agent, andexanet alfa (brand name of AndexXa ®) received FDA approval in early May 2018.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
